PLATFORM FOR ADVANCED CLINICAL THERAPIES (PACT)
Driving advanced therapeutics from bench to bedside
The Platform for Advanced Clinical Therapies (PACT) strives to close the gap between cutting-edge advanced therapeutics — such as RNA, gene and cell therapies — and their clinical implementation, making it possible to treat patients with the most challenging neurological and rare diseases.
Part of the Clinical Research Unit at The Neuro (Montreal Neurological Institute-Hospital) — one of Canada’s largest neurological clinical trial centres — PACT collaborates with industry sponsors and academic investigators to design, start-up and manage complex clinical trials that require specialized facilities and staff expertise.

INDUSTRY-LEADING TRIAL SITE
OUR TRACK RECORD
- Collaborations with national and international companies and key opinion leaders
- Extensive experience conducting Phase I- IV trials, including dedicated Phase 1 Unit for first in-human intervention
- Competence as a site to execute complex and versatile trials, involving advanced therapeutics
- Access to The Neuro’s OR facilities, Neurocritical Care Unit (NCCU), specialised pharmacy for preparation of advanced therapeutics and Interventional radiology expertise
- Vendor agreements with certified physiotherapists, speech pathologists, respiratory therapists and other trial-specific personnel
- Specialised nursing facility including space for overnight stays, laboratory and ethics teams
- Part of Catalis Quebec’s Fast Track Approval program
EXPERIENCE WITH:
- ASO therapies for Amyotrophic Lateral Sclerosis (ALS), Prion Disease, Huntington’s Disease, Parkinson’s Disease, Progressive Supranuclear Palsy (PSP), Spinocerebellar Ataxia (SCA2), Cerebral Amyloid Angiopathy and Alzheimer’s Disease
- siRNA therapies for Myotonic Dystrophy Type 1, FSHD, Spinocerebellar Ataxia Type 2, and Charcot-Marie Tooth Type 1A
- mRNA-based vaccine for Multiple Sclerosis
- CAR-T therapies for Multiple Sclerosis
- Viral vector-based gene therapy to slow early-onset Frontotemporal Dementia and and Myotonic Dystrophy 1
INVESTIGATOR- AND INDUSTRY-INITIATED TRIAL (I2T) MANAGEMENT
As part of the I2T Management Team at the CRU, the PACT has extensive experience in the design, development, and management of clinical trials involving advanced therapeutic products. The PACT supports start-up activities, regulatory submissions, trial implementation activities and management of clinical trials. Services are offered to both academic investigators and industry partners.
WHAT WE OFFER
- Scientific knowledge about experimental requirements for regulatory approval of trials involving advanced therapeutics
- Design and Development of clinical trials for various patient populations. This includes development and optimisation of clinical trial documents including clinical trial protocol, Investigator’s Brochure, Informed Consent Forms and submissions to regulatory agencies.
- Operations and Management of clinical trials ranging from study start up, site start-up and clinical trialmanagement including monitoring and other quality management strategies
- Collaborations with specialised units at The Neuro such as the McConnell Brain Imaging Centre (BIC), the Early Drug Discovery Unit (EDDU), and the Open Biobank (C–BIG)
- Critical connections to external collaborators in academic and industry with expertise and infrastructure to accommodate complex clinical trial requirements

CURRENTLY IN START-UP
▪ First in-human ASO therapy for children with Schinzel-Giedion Syndrome (PI: Carl Ernst)
▪ Industry-initiated advanced therapeutic treatment for Glioblastoma
ADVANCED THERAPY DEVELOPMENT PIPELINE
Counselling investigators to identify:
▪ Relevant, targetable genetic drivers of disease
▪ Therapeutic designs and drug screening
▪ In vitro study using cell lines (e.g. patient-derived iPSC lines)
▪ In vivo validation of therapeutic efficacy in animal models
▪ GLP Toxicology in dvanced animal models including rats and NHP
▪ Pharmacokinetic Studies
▪ Pharmacodynamic Studies
▪ GMP Certified Manufacturing
▪ Documents: Clinical Protocol Design, Investigator Brochure, Manuals, etc.
▪ Trial Logistics and Budgeting
▪ Regulatory Guidance and Submissions

▪ Site Preparation and Start-up
▪ Patient Rrecruitment and Enrolment
▪ Active Trial Conduct
▪ Data analysis and Reporting
10 YEARS OF EXPERIENCE IN EARLY PHASE STUDIES
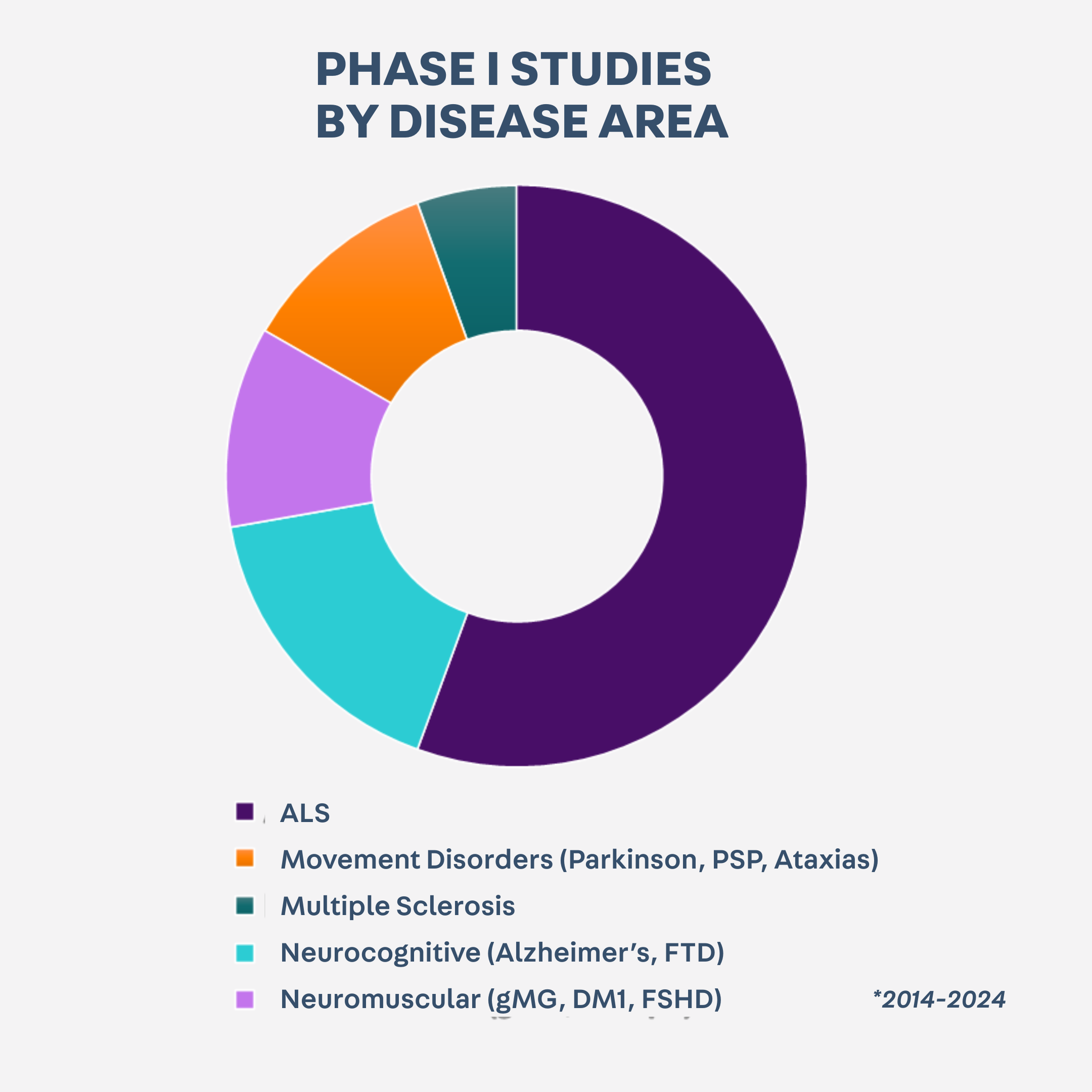
PHASE I UNIT
▪ 35 Phase I studies conducted
▪ 1st gene therapy trial in Canada for dementia
▪ Research nurses dedicated to the Unit with ICU/critical care training
▪ In-house lab team trained for Phase I trials
▪ Research pharmacy and clean room for advanced therapy products
▪ Facilities for overnight stays, access to ICU, ER
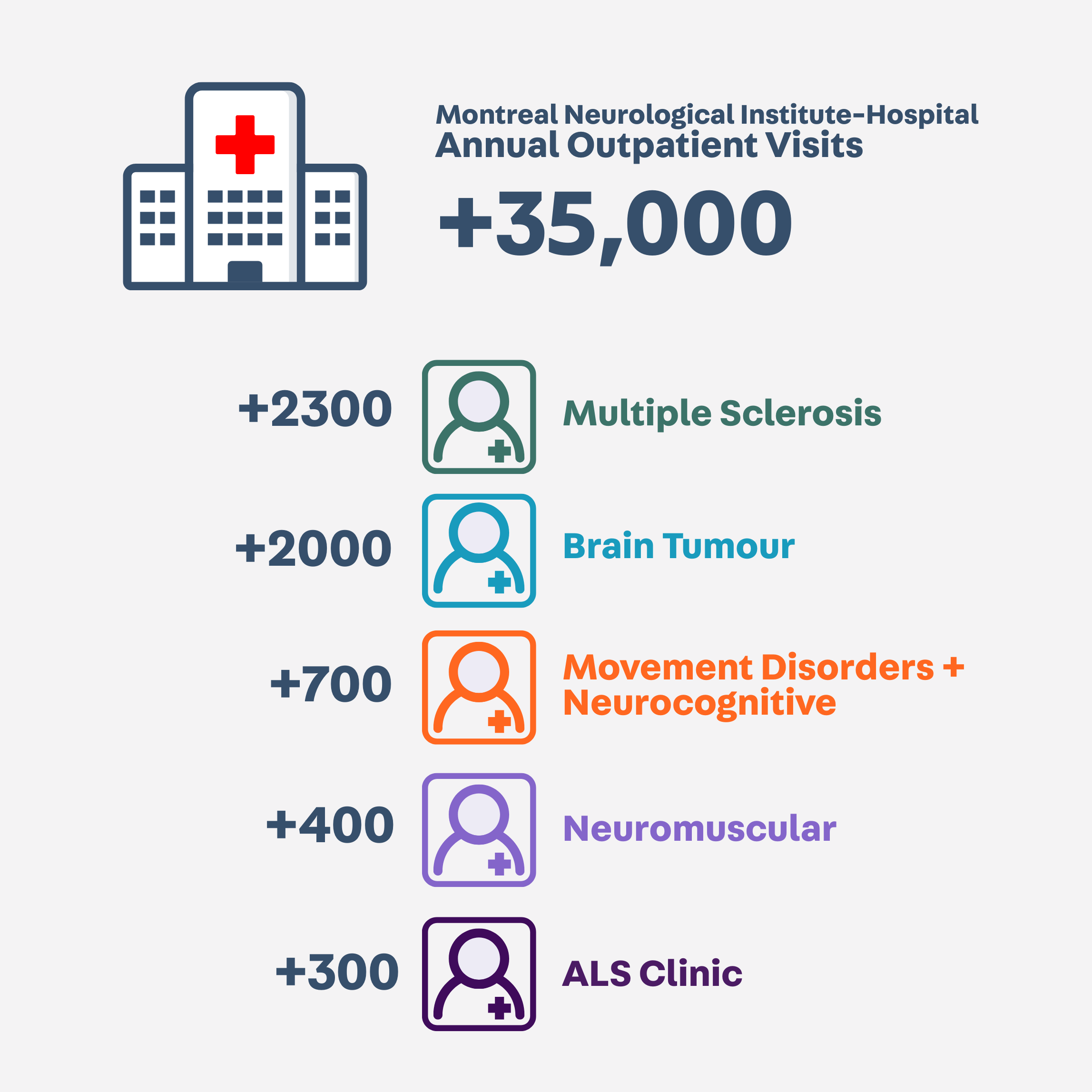
ACCESS TO LARGE NEUROLOGICAL PATIENT POPULATIONS
MANAGEMENT TEAM

ZIV GAN-OR, MD, PhD
Ziv Gan-Or’s lab works to identify targets for drug development in neurodegenerative disorders using genetic methods; understand how genetics affect disease risk, progression and response to medications; and how to apply genomics to improve clinical trials by targeting specific genetic subpopulations of patients. He works with numerous pharmaceutical companies and serves on several scientific advisory boards, to help facilitate precision medicine in PD treatment.
His work on the GBA1 gene in Parkinson’s disease (PD) helped advance the first ever phase 2 trial on a genetic subtype of PD. Other genes, that Dr. Gan-Or’s research identified as important in PD, including SMPD1, TMEM175, GALC, are currently being developed as therapeutic targets.
*Executive director, Clinical Research Unit, the Neuro
*Director, GBA1-Canada Program
*Lead, International REM-sleep Behavior Disorder Genomics Consortium
*Lead, Clinical Genetics and Gene Discovery team of the Canadian Consortium for Neurodegeneration in Aging (CCNA)
*Lead, Genetics Core of the North American Prodromal Synucleinopathy (NAPS) consortium

MASSIMO PANDOLFO, MD
Prior to coming to McGill, Dr. Massimo Pandolfo was Chief of Neurology at Erasme Hospital and Professor of Neurology at the Université Libre de Bruxelles (ULB), where he was also Director of the Laboratory of Experimental Neurology.
His research interests focus on neurogenetics. In 1996, an international collaboration led by Dr. Pandolfo was the first to identify the Friedreich’s ataxia (FRDA) gene.
He has since contributed to the study of the molecular pathogenesis of the disease, to the development of therapeutics, and to its clinical characterization. He has been the coordinator of EFACTS (European Friedreich’s Ataxia Consortium for Translational Studies) and is currently on the Board of Directors of the Ataxia Charlevoix-Saguenay Foundation.
In addition, he has contributed to the study of several monogenic epilepsies and participated in genetic studies on common epilepsies and on epilepsy pharmacogenetics.

THERESA DEGENHARD, PhD
Theresa Degenhard, PhD is a neuroscientist and project lead in the CRU’s Investigator- and Industry-Initiated Trial (I2T) Management Team, facilitating the design, start-up and management of clinical trials involving advanced therapeutics such as RNA, gene and cell therapies.
She completed her doctoral thesis with Dr. Kevin Petrecca in the Brain Tumour Research Centre at The Neuro. Her research focussed on the diversity of brain cancers using single cell technologies, as well as cell and tissue work to identify new treatment opportunities for brain cancer patients without treatment options.
A graduate of the John Molson School of Business’s Life Science Entrepreneurship Development Program, she is well versed in balancing the scientific goals of a project with the funding and regulatory requirements to translate basic research into impactful therapeutic interventions.
She enjoys driving impactful projects to support patients, caregivers, researchers and industries to create space for successful, patient-accessible medical innovation.
