I2T
INVESTIGATOR + INDUSTRY INITIATED TRIAL MANAGEMENT TEAM
END-TO-END CLINICAL TRIAL MANAGEMENT
The I2T Management Team at the Neuro CRU functions as an academic Contract Research Organization (CRO) with extensive experience in the design, development, and management of clinical trials for neurological indications.
The team supports start-up activities, regulatory submissions, trial implementation, and the management of active clinical trials. Services are available to both academic investigators and industry partners.
WHAT WE OFFER
- Scientific knowledge about experimental requirements, outcome measure selection, and compliance for regulatory approval of clinical trials.
- Design and Development of clinical trials for various patient populations. This includes development and optimisation of clinical trial documents including the Study Protocol, Investigator’s Brochure, Informed Consent Forms and submissions to regulatory agencies.
- Operations Management of clinical trials ranging from study start-up, site start-up, study monitoring, and other oversight of trial conduct.
- Collaborationswith specialised units at The Neuro such as the McConnell Brain Imaging Centre (BIC), the Early Drug Discovery Unit (EDDU), and the Open Biobank (C–BIG).
- Critical Connections to external collaborators in academia and industry with expertise and infrastructure to accommodate complex clinical trial requirements.
OUR PORTFOLIO
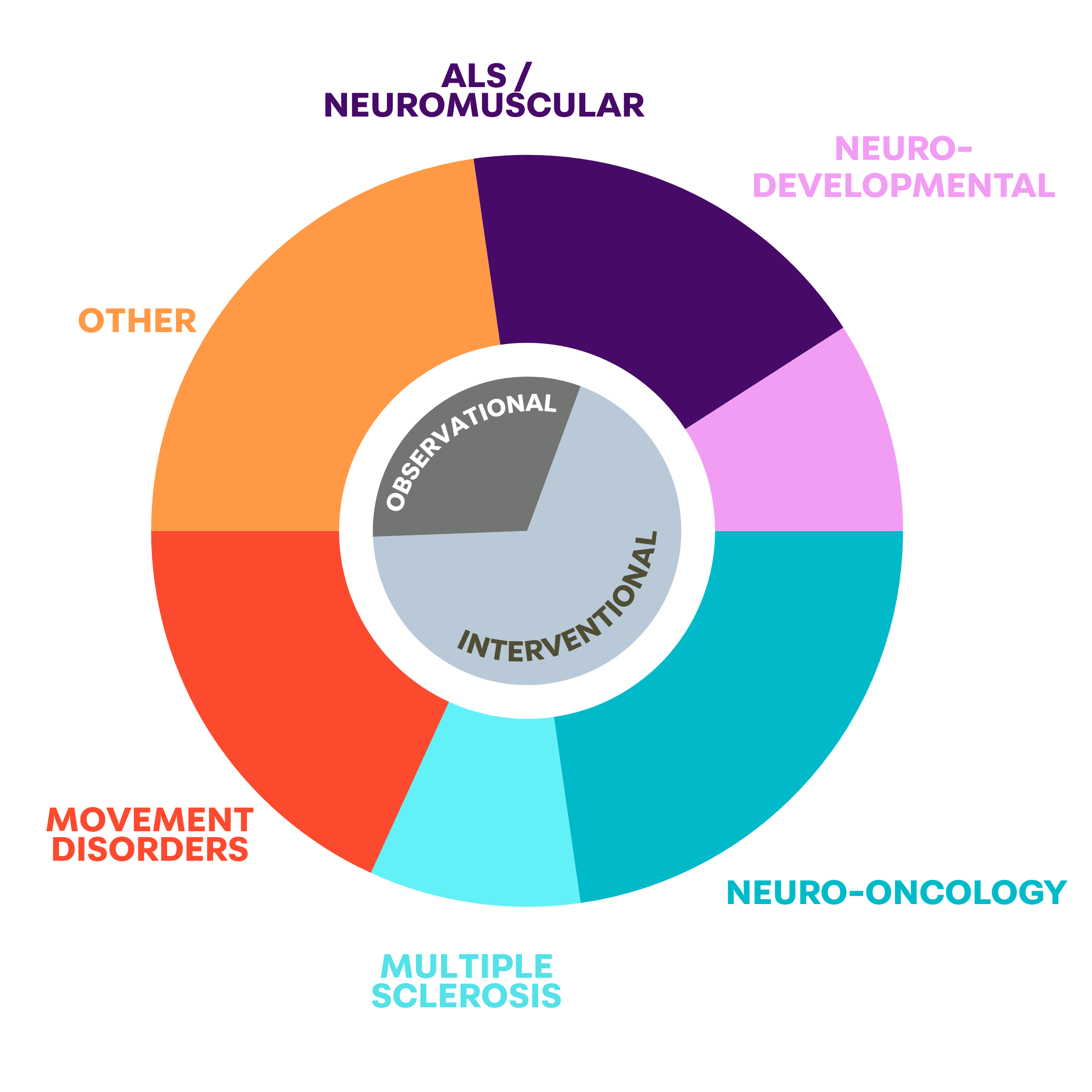
HIGHLIGHTS
Overseeing 14 studies in different stages of development or implementation:
▪ Phase 1 study for recurrent glioblastoma using an advanced therapeutic delivered directly into the brain.
▪4 Neuro-oncology trials aiming to advance the clinical diagnosis and treatment of meningioma, astrocytoma, and oligodendroglioma
CLINICAL TRIAL LIFECYCLE
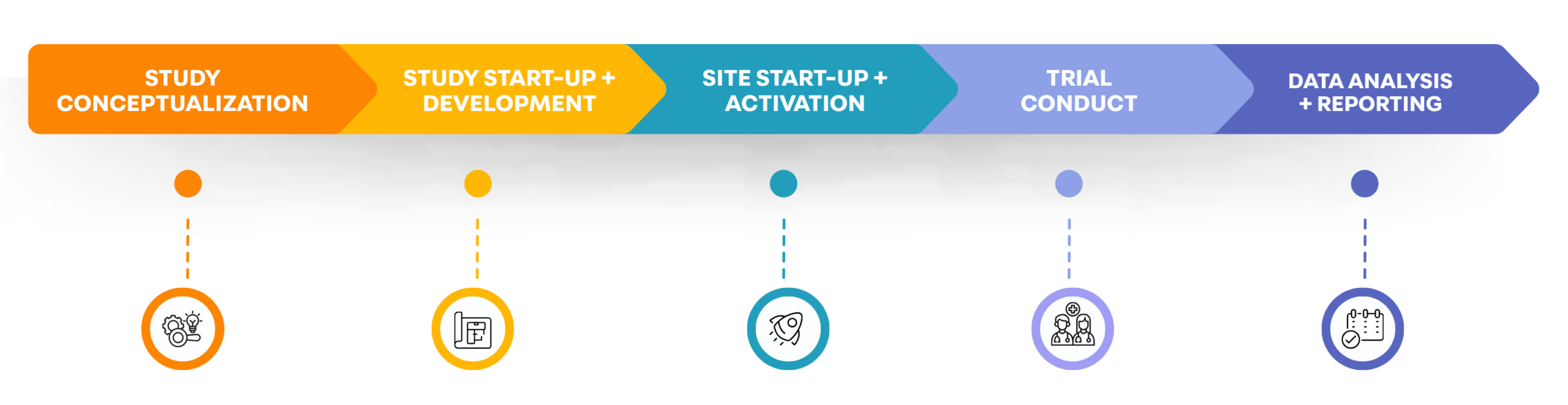
KEY CONSIDERATIONS FOR CLINICAL TRIAL SPONSORS
Trial Design and Protocol Writing
Management Contracts, Site Contracts and Vendor Contracts
Funding and Budgeting
Assembly of all Trial Documents
Health Canada Approval
Equipment, Personnel, Trial Supplies
Randomization, Blinding, Study Monitoring, Electronic Data Capture (EDC)
Data Ownership, Data Sharing, Publication Rights, Biobanking
Supply / Manufacture of Investigational Product
Post-trial Reporting Obligations to Stakeholders
ADVANTAGES OF AN ACADEMIC CRO
Deep Scientific Knowledge
Academic CROs are typically embedded in institutions with leading researchers, which provides:
- Deep therapeutic-area expertise
- Access to key opinion leaders (KOLs)
- Valuable input into study design and methodology
This can improve the scientific quality of protocols.
Strong Clinical Expertise
By being embedded in a leading neurological hospital, trials are led by clinicians with extensive disease specific experience, resulting in:
- Increased protocol compliance
- Improved site feasibility
- Expertise in outcome measure selection
This can improve the quality of endpoints.
Enhanced Cost Efficiency
Academic CROs are less expensive than large commercial CROs, particularly for:
- Investigator-initiated trials
- Early-phase or proof-of-concept studies
- Grant-funded research
They may operate with lower overhead and more flexible budgeting.
Improved Sponsor Oversight
Academic CROs usually have robust infrastructure such as:
- Institutional Review Boards (IRBs) / Research Ethics Boards
- Experienced regulatory teams
This can enhance ethical oversight, regulatory compliance, as well as protocol compliance by the site.
Access to Specialized Patient Populations
Because they are located in academic hospitals or research networks, academic CROs have:
- Better access to rare disease populations
- Specialized clinics and referral networks
- Established clinical registries and cohorts
This can make patient recruitment more efficient for certain studies.
I2T MANAGEMENT TEAM

NISHA PULIMOOD, PhD | Team Lead
Nisha Pulimood is a neuroscientist and clinical research leader with expertise spanning molecular neuroscience, stem cell-based disease modeling, and clinical trial management for rare neurological disorders.
As Team Lead for the I2T Management Team, she oversees grant development, protocol design, regulatory submissions, and site operations across Canada. Her dual background in academic and clinical research enables her to drive projects that bridge discovery science with patient-centered clinical trials.

THERESA DEGENHARD, PhD | Project Lead
Theresa Degenhard is a neuroscientist and a Project Lead in the Investigator-Initiated Trials team, driving the design, start-up, and management of clinical trials. She also manages the Platform for Advanced Clinical Therapies (PACT) at the Neuro CRU, which supports investigators in advancing late-stage preclinical therapeutics—such as RNA, gene, and cell therapies—into clinical trials.
A graduate of the John Molson School of Business’s Life Science Entrepreneurship Development Program, Theresa combines scientific insight with business acumen, aligning research goals with funding and regulatory requirements to accelerate the translation of discovery into impactful therapies.

MICHAEL PILEGGI, MSc | Project Lead
Michael Pileggi is a clinical research specialist and a Project Lead with the I2T Management Team. He has expertise in the design and execution of both preclinical and clinical research in neurobiology, neurocognitive, neuromuscular and immune-mediated indications.
As a clinical research coordinator and team lead at the Neuro CRU, Michael oversaw multidisciplinary teams supporting more than 45 active trials and served as the primary coordinator on 20 studies.
His experience spans trial feasibility, regulatory oversight, and end‑to‑end study operations, with a strong record of rebuilding teams, accelerating start‑up timelines, and sustaining high standards of GCP compliance across diverse sponsors and platforms.
